|
|
| Pronunciation | (TAN-təl-əm) |
|---|
| Appearance | gray blue |
|---|
| Standard atomic weight (Ar, standard) | 180.94788(2) |
|---|
|
|
| Atomic number (Z) | 73 |
|---|
| Group | group 5 |
|---|
| Period | period 6 |
|---|
| Block | d-block |
|---|
| Element category | transition metal |
|---|
| Electron configuration | [Xe] 4f14 5d3 6s2 |
|---|
Electrons per shell | 2, 8, 18, 32, 11, 2 |
|---|
|
| Phase at STP | Ta: Solid |
|---|
| Melting point | 3290 K (3017 °C, 5463 °F) |
|---|
| Boiling point | 5731 K (5458 °C, 9856 °F) |
|---|
| Density (near r.t.) | 16.69 g/cm3 |
|---|
| when liquid (at m.p.) | 15 g/cm3 |
|---|
| Heat of fusion | 36.57 kJ/mol |
|---|
| Heat of vaporization | 753 kJ/mol |
|---|
| Molar heat capacity | 25.36 J/(mol·K) |
|---|
Vapor pressure| P (Pa) | 1 | 10 | 100 | 1 k | 10 k | 100 k |
|---|
| at T (K) | 3297 | 3597 | 3957 | 4395 | 4939 | 5634 |
|---|
|
|
| Oxidation states | −3, −1, +1, +2, +3, +4, +5 (a mildly acidic oxide) |
|---|
| Electronegativity | Pauling scale: 1.5 |
|---|
| Ionization energies | - 1st: 761 kJ/mol
- 2nd: 1500 kJ/mol
-
|
|---|
| Atomic radius | empirical: 146 pm |
|---|
| Covalent radius | 170±8 pm |
|---|
| Spectral lines of tantalum |
|
| Natural occurrence | Ta: Primordial |
|---|
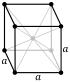
| Crystal structure | body-centered cubic (bcc)
α-Ta |
|---|
| Crystal structure | tetragonal
β-Ta |
|---|
| Speed of sound thin rod | 3400 m/s (at 20 °C) |
|---|
| Thermal expansion | 6.3 µm/(m·K) (at 25 °C) |
|---|
| Thermal conductivity | 57.5 W/(m·K) |
|---|
| Electrical resistivity | 131 nΩ·m (at 20 °C) |
|---|
| Magnetic ordering | paramagnetic |
|---|
| Magnetic susceptibility | +154.0·10−6 cm3/mol (293 K) |
|---|
| Young's modulus | 186 GPa |
|---|
| Shear modulus | 69 GPa |
|---|
| Bulk modulus | 200 GPa |
|---|
| Poisson ratio | 0.34 |
|---|
| Mohs hardness | 6.5 |
|---|
| Vickers hardness | 870–1200 MPa |
|---|
| Brinell hardness | 440–3430 MPa |
|---|
| CAS Number | 7440-25-7 |
|---|
|
| Discovery | Anders Gustaf Ekeberg (1802) |
|---|
| Recognized as a distinct element by | Heinrich Rose (1844) |
|---|
|
|
| | references |