
|
| Methane | |
|---|---|
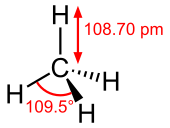 | |
| Other names |
|
| Identifiers | |
| CAS number | |
| PubChem | |
| EC number | 200-812-7 |
| KEGG | C01438 |
| MeSH | |
| ChEBI | CHEBI:16183 |
| RTECS number | PA1490000 |
| SMILES | C |
| Beilstein Reference | 1718732 |
| Gmelin Reference | 59 |
| 3DMet | B01453 |
| Properties | |
| Molecular formula | CH4 |
| Molar mass | 16.04 g mol-1 |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | |
| Melting point | -182 °C, 90.7 K, -296 °F |
| Boiling point | |
| Solubility in water | 22.7 mg·L−1 |
| Solubility | Soluble in ethanol, diethyl ether, benzene, toluene, methanol, acetone and insoluble in water |
| log P | 1.09 |
| kH | 14 nmol·Pa−1·kg−1 |
| −12.2×10−6 cm3·mol−1 | |
| Structure | |
| Td | |
| Molecular shape | Tetrahedron |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std enthalpy of formation ΔfH | −74.87 kJ·mol−1 |
| Std enthalpy of combustion ΔcH | −891.1 to −890.3 kJ·mol−1 |
| Standard molar entropy S | 186.25 J·(K·mol)−1 |
| Specific heat capacity, C | 35.69 J·(K·mol)−1 |
| Hazards | |
| NFPA 704 | |
| Explosive limits | 4.4–17% |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Methane is an organic compound with the chemical formula CH4. It is an alkane with one carbon atom. It is often found as the main part of natural gas. Methane is a greenhouse gas 23 times more effective than carbon dioxide. It is also less stable and slowly oxidates by oxygen to carbon dioxide and water.
view more...