
|
| Phosphate | |
|---|---|
 | |
| Identifiers | |
| CAS number | |
| PubChem | |
| MeSH | |
| ChEBI | CHEBI:18367 |
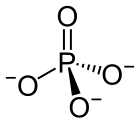
| SMILES | [O-]P([O-])([O-])=O |
| Beilstein Reference | 3903772 |
| Gmelin Reference | 1997 |
| Properties | |
| Molecular formula | PO3− 4 |
| Molar mass | 94.9714 g mol−1 |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
A phosphate is a salt of phosphoric acid. Phosphates are important in biochemistry.Phosphates have the formula PO43- and a molar mass of 94.973 g/mol. An example of a phosphate is sodium phosphate. Three different types of phosphates are known. They are orthophosphate, PO43-; metaphosphate, PO32-; and pyrophosphate, P2O73-. They have a combining power of 3.
view more...